Zoom Details:
https://ubc.zoom.us/j/65272630292?pwd=bytOVU5LRkpYY0tOdlpwQXhJV28xdz09
- Meeting ID: 652 7263 0292
- Passcode: 686756
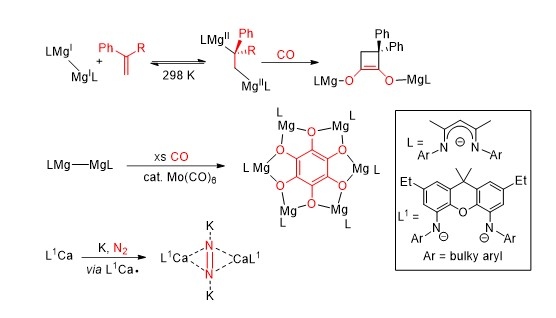
Since the synthesis of the first stable magnesium(I) compounds was achieved in 2007, the unique properties that these species possess has lent them to use as versatile reducing agents in both organic and inorganic synthetic protocols.[1] The products of such reactions are often inaccessible using more classical reducing agents. We have recently developed several classes of highly activated magnesium(I) and "masked" magnesium(I or 0) systems, and have shown these to be markedly more reactive than their established counterparts. In this lecture it will be shown that these, and related heavier alkaline earth, compounds are powerful reagents for the "transition metal-like" activation of catalytically relevant small molecules and inert arenes (e.g. CO, H2, C2H4, N2, C6H6) (Figure 1).[2-6] In several cases, small molecule activations are redox reversible under mild conditions, which gives hope for the eventual incorporation of cheap, non-toxic magnesium(I) (and heavier group 2 metal) compounds into catalytic cycles, normally requiring expensive and toxic late transition metal complexes to proceed.
REFERENCES
[1] Jones, C. Nat. Rev. Chem., 2017, 1, 0059.
[2] Boutland, A. J.; Carroll, A.; Lamsfus, C. A.; Stasch, A.; Maron, L.; Jones, C., J. Am. Chem. Soc., 2017, 139, 18190.
[3] Yuvaraj, K.; Douair, I.; Paparo, A.; Maron, L.; Jones, C. J. Am. Chem. Soc., 2019, 141, 8764.
[4] Paparo, A.; Yuvaraj, K.; Matthews, A. J. R.; Douair, I.; Maron, L.; Jones, C., Angew. Chem. Int., 2021, 60, 630.
[5] Jones, D.D.L.; Douairm I.; Maron, L.; Jones, C., Angew. Chem. Int., 2021, 60, 7087.
[6] Mondal, R.; Yuvaraj, K.; Rajeshkumar, T.; Maron, L.; Jones, C.; Chem. Commun., 2022, 58, 12665.
